Metal-On-Metal Hip Implants
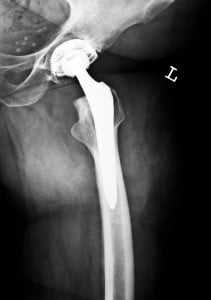
Some manufacturers have made these replacement parts out of metal. Unfortunately, this has created many dangerous complications. At Roberts & Roberts, our experienced product liability attorneys are assisting patients who have been injured by metal-on-metal hip replacements or metal-on-metal hip implants.
Information about Metal-on-Metal Hip Implants
During a hip resurfacing, physicians fit a cap into the femoral head (the top of the femur), and an artificial cup is placed inside of the socket (depression) in the pelvis that the femur slides into. The cup may be screwed in to the pelvis or can actually be made to grow into and become a part of the bones through the use of a material that promotes bone regeneration.
The cap and the cup can be made of a variety of materials, but in the mid-2000s, some manufacturers began aggressively marketing metal components for both.
For patients who undergo a total hip replacement, the femoral head is removed and the inside of the femur bone is carved out. A stem is put inside of the femur, which is topped by a new artificial head (a whole new joint). A cup is then placed inside of the socket in the pelvis for the new artificial femur head to fit into.
Again, a variety of materials can be used in hip replacements, but metal-on-metal became a popular and heavily marketed choice in the mid-2000s.For a long time, polyethylene-lined replacement cups were more commonly used to line the socket in the pelvis, while femoral heads were made of either metal or ceramic. Many were dissatisfied with these polyethylene or ceramic solutions, since the liner often wore out and needed to be replaced and the ceramic components could break or chip, especially in those who were active.
In the 2000s, metal-on-metal hip joints and resurfacing materials were marketed as a longer-lasting solution and as a better choice for younger and more active people. Many manufacturers began making these metal-on-metal products at this time and had them approved under a special set of FDA rules. The FDA’s 510(k) program allows for fast-track approval of medical devices that are substantially similar to those that have already been approved.
The marketing by the new manufacturers worked, and metal-on-metal products soon became very common. In fact, an article in the British Medical Journal in 2011 indicated that metal-on-metal products were used in as many as 10 percent of total hip operations worldwide between 2006 and 2010.
Complications from Metal-on-Metal Hip Implants and Hip Replacements
Patients with metal-on-metal hip replacements or resurfacing may experience a number of potential complications. Problems can include:
- Aseptic loosening — This simply means that the components implanted as part of the hip replacement (most commonly the acetabular cup) start to come loose. This can cause disease, pain, and infection, and must be corrected surgically.
- Metallosis — The metal hip components may begin to degrade, leaving metal debris. Metal ions may also begin to build up in the body from the metal components. This can lead to a build-up of metal in the blood and can cause serious complications, including a pseudo-tumor (a tumor-like collection of metal stained fluid), degradation of the bone and tissue death. Removal of the metal components and any remaining debris is required and must be performed surgically.
- Osteolysis — When the metal hip components release metal ions into the body, bone loss can begin to occur. This can compound other problems, especially in cases where the new hip joint and socket cap were held in place through bone growth. When the bone loss occurs, the metal components in the hip start to come loose. Surgery is the only corrective solution.
- Neurological damage — Chromium and cobalt ions in the body from the metal components can cause neurological problems over time. Symptoms include a taste of metal in the mouth that lingers, headaches, memory difficulties and heart problems.
Many patients end up having to undergo additional surgery to try to restore their health. Arthritis Today indicates that around 1 in 8 patients will require corrective surgery within five years after an original hip replacement or resurfacing.
In January 2012, a U.S. Library of Medicine article indicated that the replacement rate for certain brands of metal-on-metal devices had an even higher replacement rate, with up to 15 percent of patients needing a corrective surgery.
Dangers of Metal-on-Metal Hip Implant and Replacement Devices
Many medical studies have confirmed that these problems can develop and are linked to metal-on-metal hip replacement or resurfacing products, including a report in British Medical Journal (BMJ).
Public health agencies throughout the world have also expressed concern. According to the FDA, for example:
- The United Kingdom’s regulatory authority issued a warning in April 2010 along with specific follow-up recommendations for patients, including blood testing and imaging for patients experiencing potential complications. Updates were published in February and June 2012 with more information.
- Health Canada issued a warning to Canadian orthopaedic surgeons of potential complications in May 2012.
- Australia’s regulatory agency, the Therapeutic Goods Administration, published safety information and warnings in September 2012.
- The U.S. FDA, which the New York Times reports first received complaints as early as 2008, has issued several safety warnings and in 2012 convened the Orthopaedic and Rehabilitation Devices Panel to delve more deeply into potential risks and complications.
Some manufacturers have also recognized the dangers of their unsafe products. In August 2010, for example, DePuy voluntarily recalled its ASR™ XL Acetabular Hip System and its DePuy ASR™ Hip Resurfacing System. The British Medical Journal indicated that DePuy likely knew for a long time about the dangers of its products, as the company had an internal memo raising concerns about the potential damage due to the release of ions and about a possible cancer risk created by the breakdown of the metal on its replacement hip joints.
Problems from a Metal-on-Metal Hip Replacement or Implant? Our Lawyers Can Help
If you or a loved one has suffered complications related to a metal-on-metal hip implant or a metal-on-metal hip replacement, you could be entitled to compensation.
The defective medical device attorneys at Roberts & Roberts are currently investigating claims related to hip implants and hip replacements. For a free, confidential review of your claim, call us today at 903-597-6000 or contact us online. The call costs you nothing … It could mean everything.
Sources:







